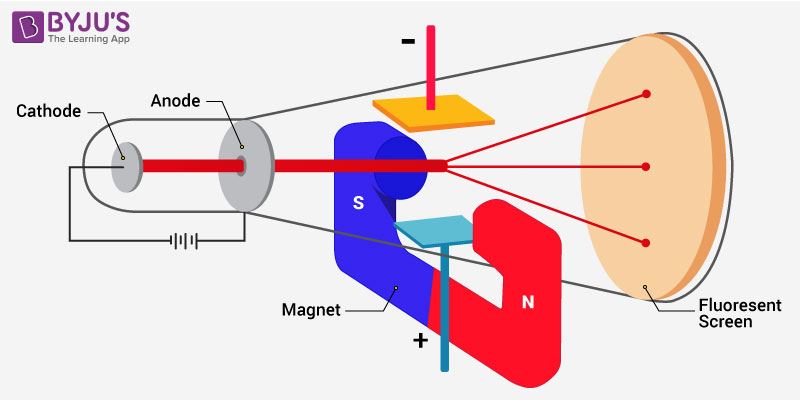
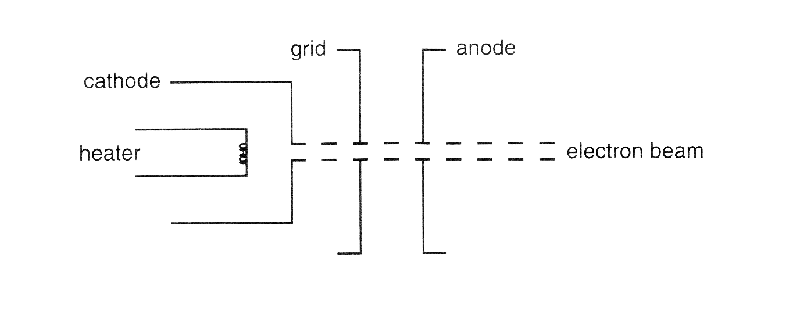
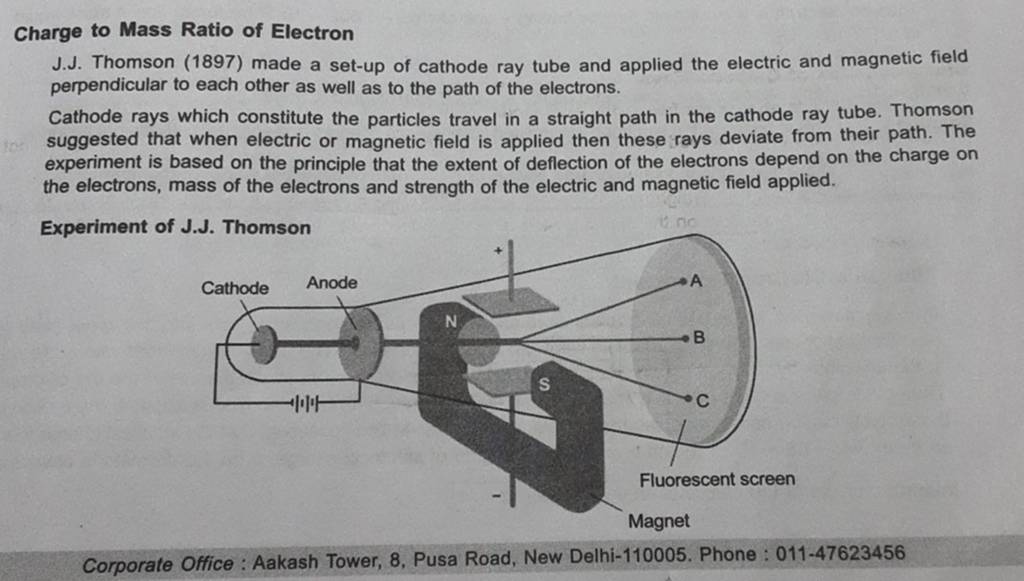
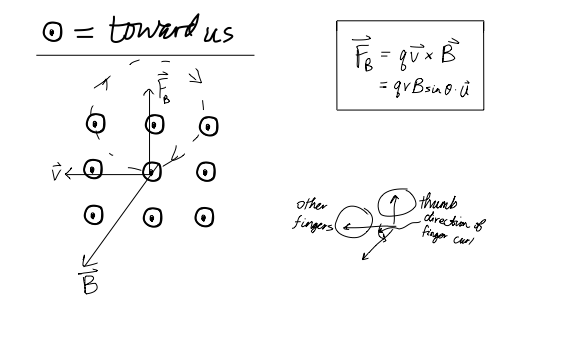
The charge to mass ratio of electron is found to be:1.602times 10^{-19} kg^{-1}1.925times 10^{-12} kg^{-1}1.758times 10^{11} kg^{-1}1.869times 10^{13} kg^{-1}

J.J. Thomson's experiment and the charge-to-mass ratio of the electron - WAEC TUTORIALS - INSIDE CHEMISTRY

Cathode Anode Fluorescent Magnet The apparatus to determine the charge to the mass ratio of electron Fig. 2.2