Concentration. Concentration Particles per volume Can be in grams per litre but chemists usually express concentration in moles per litre This are related. - ppt download
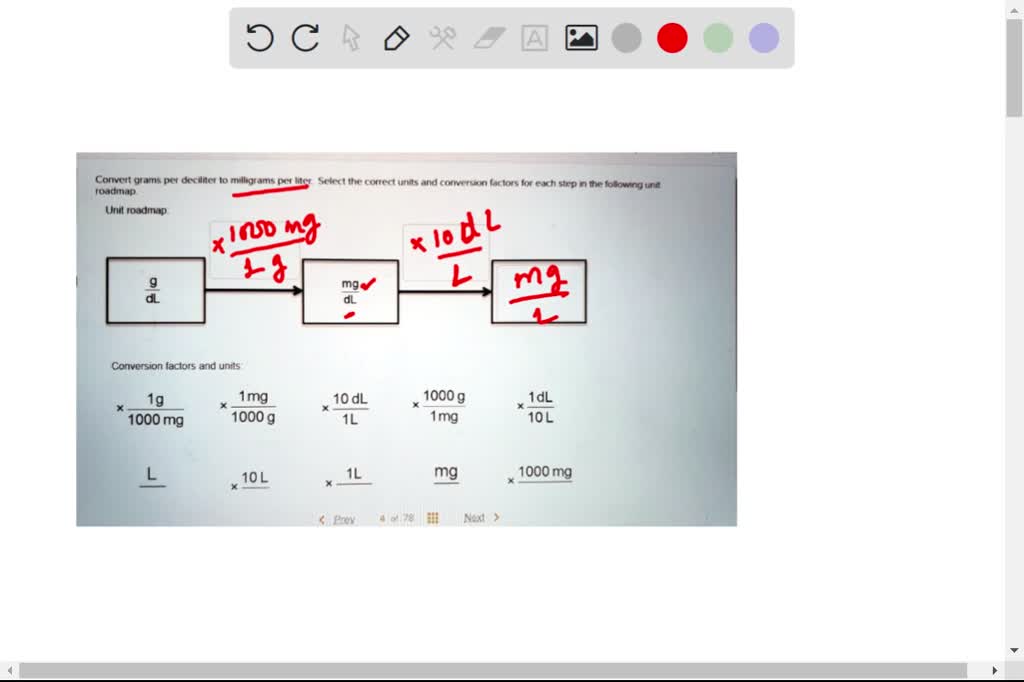
SOLVED: Convert grams per deciliter to milligrams per liter. Select the correct units and conversion factors for each step in the following unit roadmap. Conversion factors and units: 1 g = 1000
What is the solubility of nitrogen gas, in grams per liter at 25°C ? Answer in a complete [Chemistry]
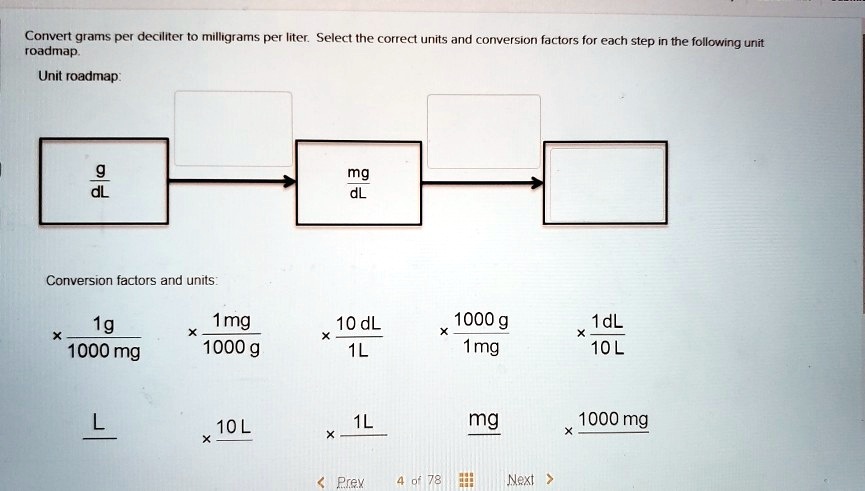
SOLVED: Convert grams per deciliter to milligrams per liter. Select the correct units and conversion factors for each step in the following unit roadmap. Conversion factors and units: 1 g = 1000

SOLVED: Problem #2: Calculate the density in grams per liter for the following gases at 0°C and 1 atm. a. NH3 b. C2H2 c. He d. H2 Problem #3: Calculate the volume

SOLVED: Calculate the solubility of each of the following compounds in moles per liter and grams per liter. (Ignore any acid-base properties.) a. A g3 P O4, Ks p=1.8 ×10^-18 b. CaCO3,

The amount of salt, in grams, in a solution is proportional to the volume, in liters, of the solution as - brainly.com